The Georgia Institute of Technology will play a key role in a new public-private partnership to help establish best practices and eventual industry-wide standards for the production of therapies using living cells to treat a range of conditions.
The new partnership aims to advance techniques to process, measure and analyze cell, gene, tissue-engineered, and regenerative medicine products, as well as cell-based drug discovery products.
“We are poised to make a significant impact in how cells and regenerative medicine products are manufactured across the world through this new strategic partnership,” said Krishnendu Roy, Robert A. Milton Chair and professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University.
Roy will serve as one of the Charter Board Members for the new International Standards Coordinating Body (SCB), which was recently established in coordination with National Institute of Standards and Technology (NIST) and Alliance for Regenerative Medicine (ARM).
“Georgia Tech has already taken a leadership role in development of best practices and analytical standards that will impact biomanufacturing and innovation through the national roadmap on cell manufacturing developed by the National Cell Manufacturing Consortium (NCMC) and the newly established Marcus Center for Therapeutic Cell Characterization and Manufacturing (MC3M),” Roy said.
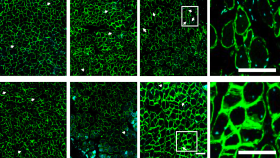
Compared to traditional pharmaceuticals, which are made primarily through chemical processes, biological medicines, also known as biopharmaceuticals, are much more challenging to produce. With medicines like cell-therapies, gene therapies and engineered tissues, finding ways to produce larger quantities at a time while ensuring high-quality and safety is a key challenge.
“The SCB’s long-term mission is to efficiently and effectively support sector standards development to accelerate product development and scalability, and streamline regulatory submission review and approval,” said Robert A. Preti, chairman of ARM and president of cell manufacturing industry partner PCT, a Caladrius Company.
The new standards initiative comes on the heels of the establishment of two National Manufacturing Innovation Institutes announced last December. One, the Institutes for Innovation in Manufacturing Biopharmaceuticals (NIIMBL), which the U.S. Department of Commerce is supporting with a five-year, $70 million grant is a consortium of more than 150 companies, academic institutions and other organizations focused towards working on improving the way biopharmaceuticals are produced, with a goal of bringing down costs and finding ways to get the drugs into the hands of clinicians and patients faster. The second, Advanced Regenerative Manufacturing Institute (ARMI) is funded by the Department of Defense and focuses on biofabrication of engineered tissues as replacement of damaged and diseases organs as well as for therapeutics development. Georgia Tech is poised to play key roles in these initiatives as well.
These synergistic national and international activities further establish Georgia Tech as a leading academic institution in the biopharmaceutical area, Roy said. In January 2016, Georgia Tech announced the Marcus Center as a research center devoted to developing processes and techniques to manufacture living cells. The center was made possible by a $15.7 million grant from the Atlanta-based Marcus Foundation. This center intends to work closely with NIIMBL and ARMI to further leverage these unique private-public partnerships and develop transformative technologies to bring cell-based therapies and regenerative medicines to clinic faster and at a lower cost.
Media Contact
Research News