With funding from the National Institutes of Health, Hanjoong Jo is leading research to understand a protein called HEG1, which could be a key to atherosclerosis. HEG1 appears to sense blood flow in arteries and then may turn on inflammatory signals in cells, a critical step toward the buildup of artery-clogging fats and cholesterol. (Image Courtesy: Arek Socha via Pixabay)
This is what scientists know about atherosclerosis, the buildup of artery-clogging fats and cholesterol in blood vessels: Good blood flow through vessels protects against it, while bad blood flow triggers the disease.
Hanjoong Jo and his team have been working to understand the detailed reasons why — and how — and recently found an interesting protein that could be a key.
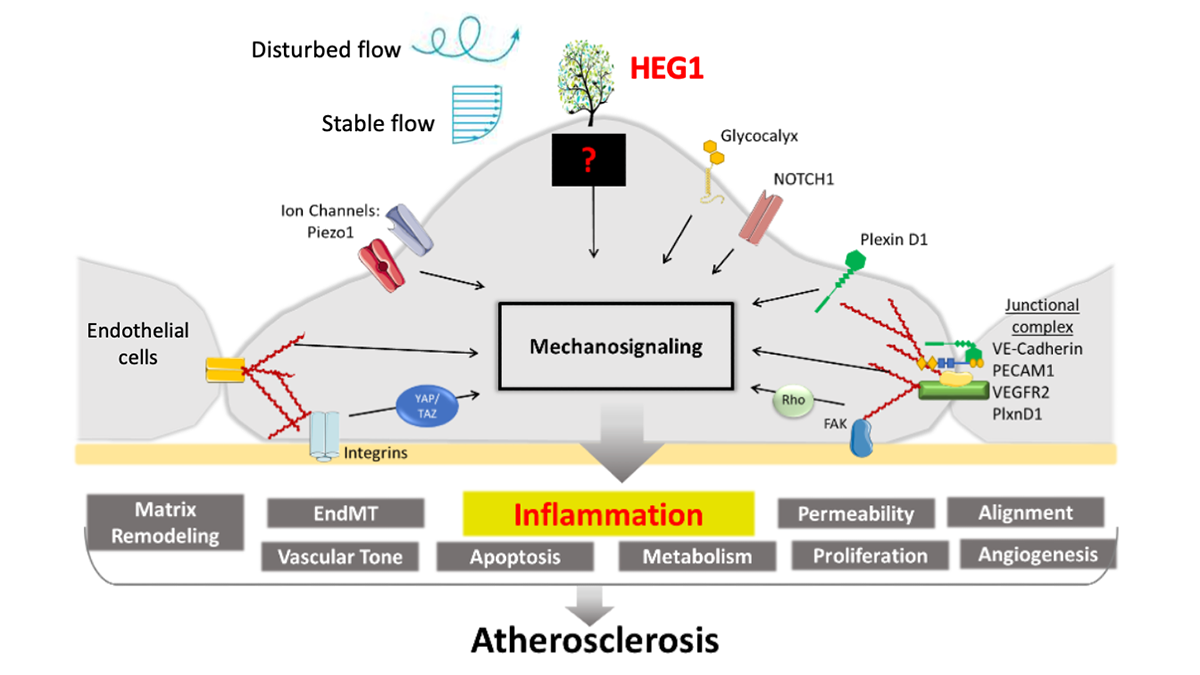
The protein, known as HEG1, is found on the endothelial cells lining our blood vessels. It appears to act as a critical blood flow sensor, keeping everything humming along as it should when blood flow is good and turning on inflammatory signals, a critical step toward atherosclerosis, when blood flow is bad.
Jo wants to know just how HEG1 works — with the hope that a deeper understanding could lead to new options for preventing or curing atherosclerotic diseases — and he has the chance with a new $2.68 million grant from the National Institutes of Health.
“We are really excited about this flow-sensitive protein, HEG1, because this could be potentially a new sensor that detects how blood is flowing in the vessel — whether it goes one way or multiple directions, at a high speed or low speed — and this flow sensor could then play a very important role in preventing or causing atherosclerosis,” said Jo, Wallace H. Coulter Distinguished Chair and Professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University. “We have shown some evidence, which allowed us to get this grant, that good flow makes more of this HEG1 gene and protein, preventing inflammation. If you have a bad flow, this potential flow sensor is reduced, causing inflammation.”
Jo described the protein produced by the HEG1 gene like a big tree with lots of branches sticking out to catch the wind as it blows. The HEG1 protein has branches jutting out from endothelial cells into arteries to catch the blood flowing by: “It is just perfectly shaped and positioned, but nobody has studied it. So, we are studying it.”
Illustration of mechanosensors and mechanosignaling in endothelial cells that line our blood vessels. Atop the cell is the tree-like HEG1 protein, which appears to act as a critical blood flow sensor in arteries. When blood flow is disturbed, this molecule could be a key to turning on inflammatory signals, a critical step toward atherosclerosis. Hanjoong Jo is leading a new National Institutes of Health-funded project to investigate the functioning of this interesting protein. (Image Courtesy: Hanjoong Jo)
Scientists have known for a while that HEG1 is important. Other researchers removed the gene in zebrafish and mice and the results were catastrophic: blood vessels — normally tightly sealed conduits — started to leak, and hearts without HEG1 grew very large and very fragile. That’s where HEG1 gets its name: It’s the “heart of glass” gene.
The question now is, does HEG1 have another crucial role in cardiovascular disease?
Jo and his team — including M.D./Ph.D. student Ian Tamargo and postdoctoral fellow Aitor Andueza — want to establish the link. If it’s there, it opens up new targets for therapies to treat atherosclerosis, a disease that can lead to heart attacks, strokes, and peripheral artery disease.
“We already know how important HEG1 is,” Jo said. “We just don't know whether flow has anything to do with it and whether it really plays a role as a sensor. And if it does act as a sensor, then how does it control the atherosclerosis-related functions? These are all unknown questions.”
In the four-year NIH project, Jo and his team will dive deeply into how HEG1 works. Extending Jo's tree analogy, researchers will trim the tree in different ways to see how cells respond. They may adjust how the roots grow — Jo said the junction between the protein outside the cell and inside of the cell looks like it could be a critical sensing area. Maybe they'll remove the tree altogether, or keep the tree in some areas but not in others. All of that will help them understand the mechanisms of HEG1's functions.
Jo’s collaborators will be key in the investigation, including several Coulter BME researchers: James Dahlman’s lab will help to develop ways to either delete or overexpress various forms of the HEG1 gene in the artery walls of mice; Cheng Zhu’s lab will help with studies to quantify how much and what types of mechanical forces can turn on or off the HEG1 molecule’s response at the single-cell level; Sung Jin Park will study calcium signaling in live cells in response to flow; and Sandeep Kumar will carry out single cell RNA sequencing studies to understand the effect of HEG1 in cells, animals, and human patient tissues with atherosclerosis. Emory cardiologist Kathy Griendling will collaborate on understanding how HEG1 signals inside the cells.
“We will really look inside the cells and explore the inflammatory response that is happening,” Jo said. “If the HEG1 proteins are sensing blood flow, what are the next steps? Are they talking to other proteins? Which ones, and how do they trigger the next events?
“Knowing that detail of the mechanisms will actually help us to figure out how to develop therapeutics.”
Latest BME News
Jo honored for his impact on science and mentorship
The department rises to the top in biomedical engineering programs for undergraduate education.
Commercialization program in Coulter BME announces project teams who will receive support to get their research to market.
Courses in the Wallace H. Coulter Department of Biomedical Engineering are being reformatted to incorporate AI and machine learning so students are prepared for a data-driven biotech sector.
Influenced by her mother's journey in engineering, Sriya Surapaneni hopes to inspire other young women in the field.
Coulter BME Professor Earns Tenure, Eyes Future of Innovation in Health and Medicine
The grant will fund the development of cutting-edge technology that could detect colorectal cancer through a simple breath test
The surgical support device landed Coulter BME its 4th consecutive win for the College of Engineering competition.